 
Divide the amount of moles you have of the known substance by the coefficient of the known substance.įor example divide 1.05 moles of MgCl2 by 1 (coefficient of MgCl2 in the balanced equation) = 1.05.Calculate Amount of Needed Substancesįinally, to calculate the amount of the other substances: To convert between moles and grams, multiply moles by the molar mass to get grams, or divide grams by the molar mass to get moles.įor example, lets say we have 100g of MgCl2 and want to convert it to the number of moles: 100/95.211 = 1.05 moles. In stoichiometric calculations, this is usually the known amount (in grams or moles) of at least one reactant or product. Moles and Grams of the Known or Needed SubstanceĮvery calculation starts with some knowns or inputs. You now have all the information needed to start doing stoichiometric calculations. 
This can be done using our molar mass calculator or manually by following our tutorial. Once you have a balanced equation, determine the molar mass of each compound. Determine the Molar Mass of Each Reactant and Product This means that for every molecule of MgCl2, you need two NaOH to form a single Mg(OH)2 molecule and two NaCl molecules. 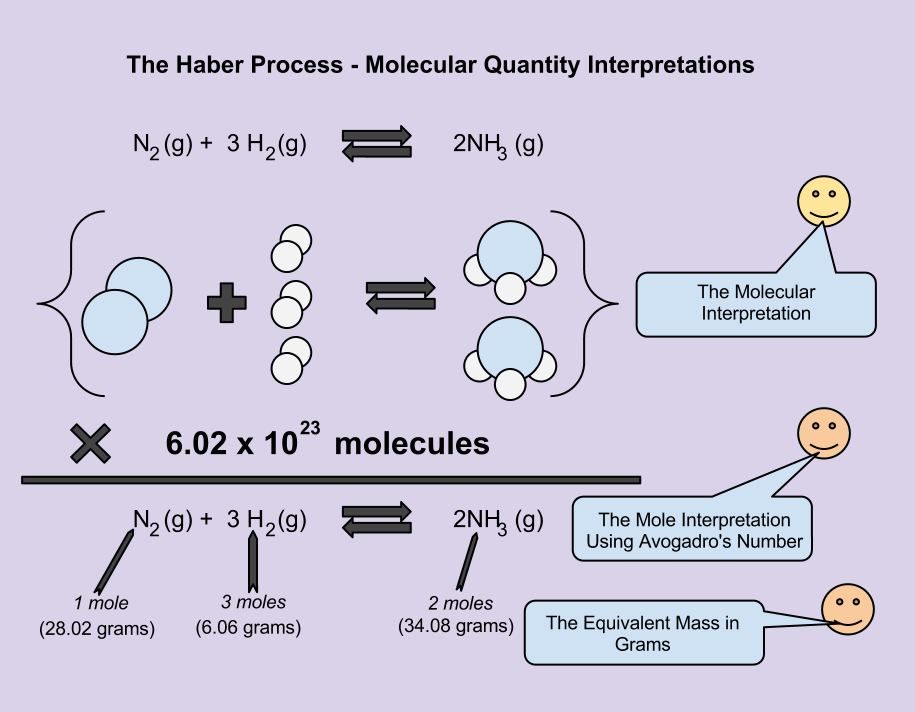
You can learn how by reading our article on balancing equations or by using ourįor example: MgCl2 + NaOH → Mg(OH)2 + NaCl is balanced to get MgCl2 + 2NaOH → Mg(OH)2 + 2NaCl. Balance the Equationīefore you can perform stoichiometric calculations, you must first balance the chemical equation. In a similar way, chemists study the proportion of substances in reactions to find out how they interact with each other. When we bake a cake, we use basic stoichiometry to determine how much of each ingredient contributes to the final product.īy adjusting the proportions of the ingredients, we can create different types of cakes. The word 'stoichiometry' is derived from the Greek words 'stoikhe' (meaning proportion or balance) and 'eikion' (meaning amount). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
